The difference between disinfect and sterilize comes down to one critical factor: the level of microbial elimination. While often used interchangeably in casual conversation, in the biohazard remediation industry, this distinction is a matter of public health and safety.
Disinfection is a process that eliminates or inactivates the vast majority of harmful microorganisms on a surface, dramatically reducing the risk of infection. Sterilization, however, is an absolute process designed to destroy all forms of microbial life, including highly resistant bacterial spores. Understanding which process is required is fundamental to ensuring a space is truly safe.
Disinfection and Sterilization: The Core Differences
When our certified technicians arrive at a scene—whether for a trauma cleanup or a large-scale contamination—knowing which protocol to implement is non-negotiable. Applying the wrong method means leaving invisible threats behind. A surface may look clean, but it can still be biologically hazardous. This decision is not a matter of preference; it is dictated by a scientific assessment of the risk level involved.
Disinfection is a cornerstone of daily safety protocols and professional biohazard remediation, but it does not guarantee a 100% kill rate. Sterilization is the definitive solution, reserved for scenarios where a complete absence of microorganisms is mandatory, such as with surgical instruments or within clean room environments.
Key Distinctions Explained
In professional and regulated settings, the precise meanings of these terms are critical. The ultimate objective is always to restore safety, which begins with applying the scientifically correct standard of care for the specific biohazards present.
Understanding the different 3 types of sanitizer solutions provides insight into the tools available for these tasks. This knowledge is fundamental to how professionals approach biohazard scenes, where standard cleaning methods are inadequate and unsafe. To understand why our work goes far beyond janitorial services, see our guide on biohazard vs. standard cleaning.
The critical takeaway is that disinfection reduces the number of pathogens to a safe level, while sterilization eliminates them entirely. This distinction forms the basis of all professional remediation and public health protocols.
At-a-Glance Comparison Disinfection vs Sterilization
To clarify these concepts, this breakdown highlights what sets disinfection and sterilization apart, including their outcomes and typical applications.
| Attribute | Disinfection | Sterilization |
|---|---|---|
| Outcome | Eliminates most pathogenic microorganisms (but not necessarily all microbial life). | Destroys all forms of microbial life, including viruses, fungi, and bacterial spores. |
| Level of Kill | Reduces microbial load to a level considered safe; it is not an absolute process. | Absolute kill; achieves a Sterility Assurance Level (SAL), typically 10⁻⁶. |
| Common Methods | Chemical agents (bleach, alcohol, quaternary ammonium compounds), UV-C light. | Physical or chemical processes (autoclaving with steam, ethylene oxide gas, radiation). |
| Typical Use | Non-critical surfaces like floors, countertops, and medical equipment that contacts intact skin. | Critical items like surgical instruments, implants, and tools entering sterile body cavities. |
This comparison underscores the significant gap between the two processes. While both are crucial for safety, their application is determined by the required level of microbial control and the associated risks.
The Science of Microbial Elimination

To fully grasp the difference between disinfect and sterilize, it is essential to understand how each method works at a microscopic level. It is not merely about wiping a surface clean; it is about applying the correct scientific principle to neutralize a specific biological threat. In the biohazard remediation profession, this choice is critical—public health depends on it.
Disinfection is a form of microscopic chemical warfare. The agents used are designed to attack and deactivate microorganisms, rendering them harmless. However, while highly effective, disinfection does not promise to eliminate everything, particularly resilient bacterial spores.
How Disinfection Works
Disinfectants target the critical functions that sustain bacteria and viruses. For instance, common agents like quaternary ammonium compounds (quats) function by disrupting the cell membrane. Once this protective barrier is breached, the cell lyses and dies. This is why quats are widely used for general cleaning in homes and non-critical areas of healthcare facilities.
More potent agents include sodium hypochlorite—the active ingredient in bleach. It works through oxidation, which aggressively degrades proteins and destroys genetic material like DNA and RNA. This process completely halts a microbe's ability to function or replicate. Its broad-spectrum efficacy makes it a primary tool in professional biohazard cleanup, but its corrosive nature demands expert handling and adherence to strict safety protocols.
One of the most critical principles in our industry is that disinfection cannot occur without thorough pre-cleaning. Organic matter like dirt, blood, or bodily fluids can shield microorganisms, rendering even the strongest disinfectants ineffective. This is a non-negotiable first step in every 360 Hazardous Cleanup protocol.
The Absolute Process of Sterilization
Sterilization operates on an absolute principle: the complete and total destruction of all microbial life, with no survivors. It employs powerful physical or chemical forces to achieve this, making it essential for environments where even a single surviving microbe could be catastrophic. This level of certainty requires specialized equipment and rigorously controlled protocols.
Several primary methods are used to achieve true sterilization, each selected based on material compatibility and specific scenarios.
- Autoclaving (Pressurized Steam): This is the gold standard for sterilization. By exposing items to high-pressure steam at extreme temperatures (typically 121°C or 250°F), it denatures all proteins and eliminates even the most resistant bacterial spores. It is the primary method hospitals use to sterilize surgical instruments.
- Ethylene Oxide (EtO) Gas: For heat-sensitive items, EtO gas provides a low-temperature sterilization solution. The gas permeates materials and disrupts microbial DNA, preventing reproduction. Because EtO gas is toxic, this process requires careful handling and specialized ventilation systems.
- Radiation: High-energy gamma rays or electron beams are used to sterilize single-use medical supplies like syringes and catheters. The radiation inflicts irreparable damage to microbial DNA, ensuring sterility without heat.
Selecting the appropriate method requires deep technical knowledge of the item's composition, the nature of the contamination, and all relevant regulatory standards. For those interested in the science that underpins our work, our guide on the processes and techniques of biohazard cleanup provides further detail.
Understanding Log Reduction and Safety Margins
To truly understand the difference between disinfecting and sterilizing, we must look beyond definitions and examine the data. In professional biohazard remediation, we measure a process's effectiveness using a scientific scale called log reduction. This quantifies precisely how many microorganisms are eliminated, providing a clear, evidence-based picture of the safety margin being created.
Each "log" on the scale represents a 90% reduction in microbes. A 1-log reduction kills 90% of microorganisms, leaving 10% behind. While this may sound significant, in high-risk environments, it is often insufficient. As you move up the scale, the standards become exponentially more stringent.
Defining Levels of Microbial Kill
The log reduction scale is exponential, not linear. Each step up represents a tenfold increase in killing power. This mathematical precision is what distinguishes professional biohazard remediation from standard cleaning and provides a framework for accurate risk assessment.
Here is a brief breakdown:
- 1-log reduction: Kills 90% of microbes.
- 2-log reduction: Kills 99% of microbes.
- 3-log reduction: Kills 99.9% of microbes.
- 6-log reduction: Kills 99.9999% of microbes. This is the established standard for critical applications, capable of reducing a colony of one million organisms to just one.
This is precisely why choosing the correct process is so important. A 3-log reduction may be acceptable for a low-risk office desk, but at a trauma scene or in a healthcare setting, anything less than a 6-log reduction could leave dangerous pathogens behind.
The Sterility Assurance Level
Sterilization elevates this concept to its absolute extreme by targeting a Sterility Assurance Level (SAL). For medical devices and other critical applications, the universally accepted standard is an SAL of 10⁻⁶. This is not an estimate but a statistical probability, signifying less than a one-in-a-million chance that a single viable microorganism remains on an item after the process is complete.
An SAL of 10⁻⁶ is more than just a high number; it’s a promise of total microbial destruction. It provides the largest possible safety margin, which is non-negotiable when dealing with invasive medical procedures or remediating highly infectious biological materials.
This represents the fundamental divide between disinfection and sterilization. Even a high-level disinfectant can achieve an impressive log reduction, but it cannot offer the statistical certainty of an SAL. For example, research on medical instruments has shown how narrow this margin can be. Even with top-tier disinfection and cleaning, the safety margin may be minimal or non-existent, depending on the initial bioburden of the item.
Comparing Safety Margins in Practice
The difference between a 99.9% kill rate (disinfection) and a 99.9999% kill rate with a one-in-a-million chance of survival (sterilization) is immense. When dealing with dangerous pathogens, that small fraction of a percentage can be the difference between a safe environment and a source of future infection. This is particularly true for highly resilient organisms, which you can learn more about in our comprehensive guide to cleaning superbugs.
At 360 Hazardous Cleanup, our protocols are built on this scientific foundation. We do not just clean a surface; we assess the unique biohazard risk and apply the correct, scientifically validated method to achieve the required log reduction. This commitment to quantifiable results is how we restore an environment to a state of true biological safety, protecting the health of all future occupants.
When Each Method Is Required by Regulations
Understanding the difference between disinfect and sterilize is not just a matter of technical accuracy—it is a critical component of regulatory compliance. Agencies such as the Centers for Disease Control and Prevention (CDC), the Occupational Safety and Health Administration (OSHA), and the Environmental Protection Agency (EPA) have established strict protocols that dictate which method must be used based on specific risks.
These are not mere suggestions; they are legally mandated standards designed to protect public health. For professionals in the biohazard remediation field, these guidelines are the bedrock of every decision made on-site, ensuring a restored environment is not just visibly clean but verifiably safe.
The Spaulding Classification System
A cornerstone of these regulatory frameworks is the Spaulding Classification, a system widely adopted in healthcare and biohazard remediation to categorize items based on their risk of causing infection. It removes guesswork by providing a clear, risk-based logic for choosing between disinfection and sterilization.
The system classifies items into one of three categories, each with its own non-negotiable protocol:
Non-Critical Items: These are surfaces that only come into contact with intact skin, such as floors, countertops, or blood pressure cuffs. Since unbroken skin acts as an effective natural barrier, these items pose the lowest risk and typically require low-level or intermediate disinfection.
Semi-Critical Items: This category includes items that contact mucous membranes or non-intact skin, like respiratory therapy equipment. These require, at minimum, high-level disinfection to eliminate all microorganisms except for high numbers of bacterial spores.
Critical Items: This is the highest-risk category, encompassing anything that enters sterile body tissue or the vascular system, such as surgical instruments, needles, or implants. The risk of infection is so severe that these items demand sterilization. There is no alternative.
This flowchart helps visualize how these risk-based decisions are made.
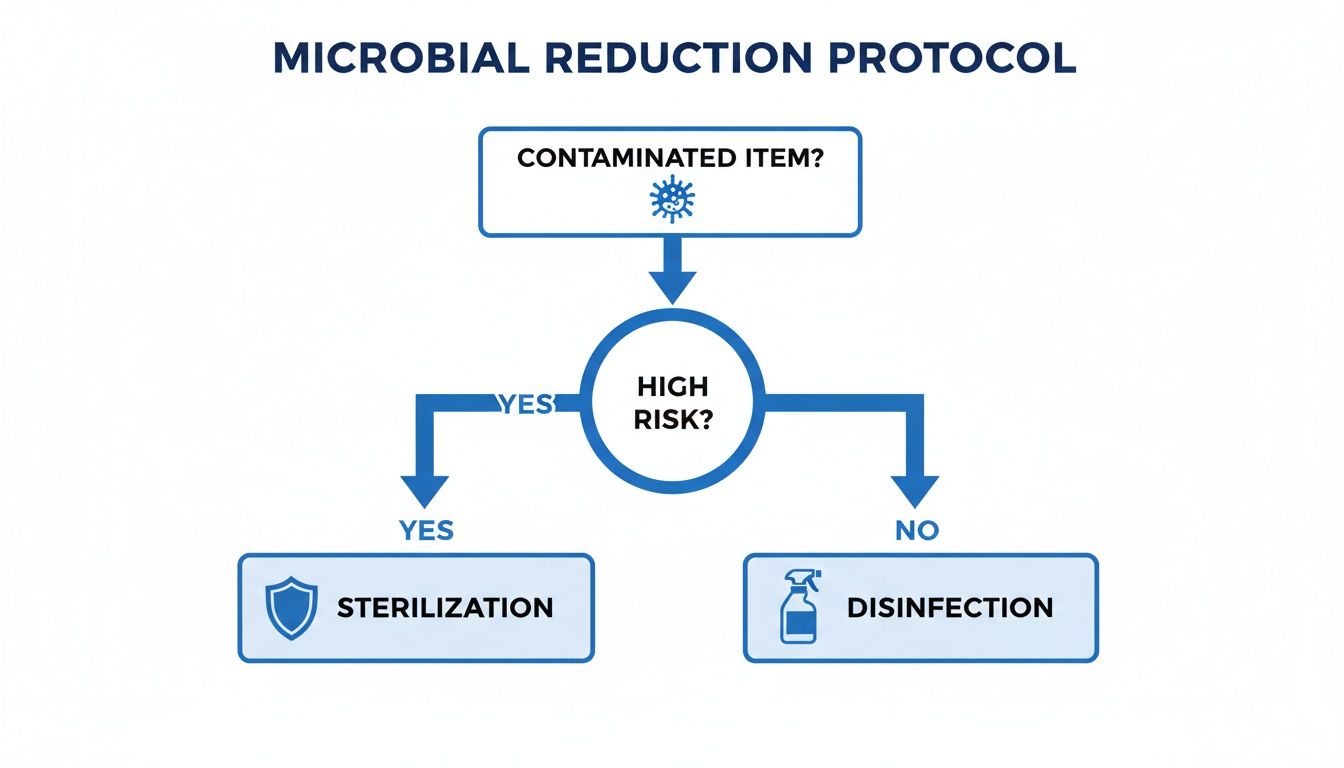
The key takeaway is that the level of potential harm dictates the necessary response, with sterilization reserved for the absolute highest-risk situations.
Regulatory Mandates in Practice
Regulatory bodies provide clear directives that leave no room for error in high-risk environments. The CDC, for instance, mandates that all critical medical and surgical devices must be fully sterilized before every use. It is a categorical rule with no exceptions for cost or convenience.
In the biohazard remediation industry, we apply the exact same logic. A blood-soiled floor is a non-critical surface, but it still requires meticulous high-level disinfection. Conversely, any potentially contaminated sharp object that could pierce the skin is handled with protocols that assume the highest possible risk, demanding sterilization-level disposal methods.
This structured, compliance-driven approach is what separates professional remediation from standard cleaning. Adherence to these standards is a core competency of every 360 Hazardous Cleanup technician. For a closer look at these safety requirements, our guide on the OSHA guidelines for biohazard cleanup provides essential information. Our protocols are designed to meet or exceed every one of these regulatory mandates, ensuring each site is restored to a state of complete biological safety.
Practical Applications in Biohazard Remediation

Knowing the textbook difference between disinfect and sterilize is one thing; applying that knowledge correctly in the field is what truly restores a space to safety after a traumatic event. At 360 Hazardous Cleanup, our protocols are never one-size-fits-all. We develop a tailored action plan based on a rigorous, on-site assessment of the specific biohazard situation.
This is where professional expertise is indispensable. Every scene, from an unattended death to a hoarding situation, presents a unique combination of contaminants, surfaces, and health risks that dictate the required level of microbial elimination. A space that appears clean is not necessarily safe. Our mission is to make it biologically safe for anyone who enters after our work is complete.
Tailoring Protocols to Real-World Scenarios
Consider a hoarding cleanup. The primary challenge is often widespread bacterial growth from decay, waste, and filth across non-critical surfaces like walls, floors, and personal belongings. In these cases, the correct protocol involves a multi-step process: removing gross filth and debris, followed by the application of broad-spectrum, EPA-registered disinfectants to eliminate pathogens from large areas.
A crime scene or trauma cleanup, however, presents a more immediate biological risk from bloodborne pathogens, demanding a more focused strategy. Our technicians are trained to identify all affected materials, contain and remove biohazardous waste according to strict regulations, and then meticulously apply high-level disinfectants to all non-porous surfaces. This process achieves the necessary log reduction to neutralize threats like HIV or Hepatitis.
The decision to disinfect versus sterilize is made on a micro-level at every job site. It’s a judgment call guided by science, experience, and an unwavering commitment to regulatory compliance, ensuring the chosen method directly counters the specific biological threat present.
Addressing Highly Resistant Organisms
Certain biohazards are notoriously difficult to eliminate and require specialized protocols. Decontaminating a space after an infectious disease outbreak, particularly one involving spore-forming bacteria, highlights the necessity of selecting the right chemical agent and adhering to precise contact times. Without a deep understanding of microbiology, it is impossible to guarantee complete inactivation.
This is especially true when dealing with Clostridioides difficile. Healthcare facilities often rely on sporicidal solutions containing 5,000 ppm chlorine for terminal cleaning of rooms where a C. diff patient was treated. Studies have shown that a 1:10 dilution of concentrated bleach is highly effective at reducing environmental contamination. You can learn more about these specific challenges in our detailed guide on C. difficile cleaning protocols.
In every scenario, our technicians methodically document the entire process, from the initial assessment to final clearance testing. This scientific approach provides peace of mind by confirming that the property is not just visually restored but biologically safe. It is this meticulous, real-world application of disinfection principles that defines professional biohazard remediation and protects the well-being of families and communities.
Why Professional Expertise Is Non-Negotiable
Knowing the textbook difference between disinfect and sterilize is one thing. Applying that knowledge correctly under the immense pressure of a real-world biohazard scene is what separates a professional from an amateur.
When it comes to biohazard remediation, the stakes could not be higher. A single misstep can create lasting health risks for future occupants. This is why engaging a certified expert is not just advisable—it is an absolute necessity for safety and compliance.
Improper cleanup attempts are fraught with risks. Using the wrong chemical concentration will fail to eliminate dangerous pathogens. Misjudging the required contact time renders a disinfectant useless. An untrained individual might even select a product that is ineffective against the specific bacteria or viruses present, leaving behind a false sense of security in a space that remains biologically contaminated.
The Hidden Dangers of DIY Cleanup
Beyond technical errors, there are significant safety and regulatory challenges that only a trained team can navigate. Proper Personal Protective Equipment (PPE) is far more than just a pair of gloves; it requires comprehensive respiratory and dermal protection to shield technicians from invisible threats that can cause serious illness.
Furthermore, handling and disposing of biohazardous waste is a tightly regulated process. Materials contaminated with blood or bodily fluids cannot be discarded in standard trash. This waste is legally classified as regulated medical waste and is subject to strict requirements for packaging, transport, and disposal to comply with federal and state laws. Failure to follow these rules can result in heavy fines and pose a significant risk to public health.
In a time of crisis and grief, the last thing a family should have to do is learn complex safety protocols and navigate regulatory red tape. A professional's job is to take on that burden, handling the entire remediation process with precision, compassion, and unwavering compliance.
The 360 Hazardous Cleanup Commitment
At 360 Hazardous Cleanup, our work is built on a foundation of scientific knowledge, regulatory expertise, and genuine empathy. We understand that we are not just cleaning a property; we are helping people through one of the most difficult times of their lives. Our technicians are highly trained in risk assessment, microbial decontamination, and regulatory compliance, ensuring every job is completed correctly and the site is restored to a verifiably safe condition.
We operate with discretion and sensitivity, using unmarked vehicles upon request and maintaining clear communication throughout the process. Our role is to manage the technical complexities so you and your family can focus on healing. From the initial assessment to coordinating with your insurance provider, we handle every detail to provide you with complete peace of mind.
Frequently Asked Questions
Understanding the distinction between disinfecting and sterilizing is critical for ensuring true biological safety. Even after explaining the science, many people have practical questions. We have compiled some of the most common ones to provide clear, straightforward answers.
These insights should help clarify any remaining confusion and demonstrate why professional intervention is essential when dealing with a hazardous situation.
Can Something Be Clean But Not Disinfected?
Absolutely. This is one of the most important concepts in our field. Cleaning is the process of removing visible dirt and grime. It is an essential first step, as organic material can shield germs from the chemical agents intended to eliminate them.
A countertop can appear spotless but still harbor millions of microscopic bacteria and viruses. In professional biohazard remediation, a site is never considered safe simply because it looks clean. It is only safe after it has been properly disinfected to meet public health and safety standards.
Is Hand Sanitizer a Disinfectant?
While they belong to the same family of germ-killers, they are not interchangeable. They are different tools for different applications. Hand sanitizers are antiseptics formulated for use on skin and are regulated by the FDA. They typically use alcohol to reduce the number of germs on your hands.
Surface disinfectants, conversely, are regulated by the EPA and formulated for hard, non-porous surfaces like floors and countertops. They are far too harsh to be used on skin. Using a surface disinfectant on your hands is dangerous, and using hand sanitizer to clean a countertop is ineffective. It is critical to use the right product for its intended purpose.
The difference between sanitizing and disinfecting comes down to kill-rate. Sanitizing lowers the number of germs to a safe level. Disinfecting eliminates nearly 100% of the specific pathogens listed on the product's label, as long as you follow the required contact time.
When Is Sterilization Necessary in a Home?
For the vast majority of homeowners, the answer is simple: almost never. A routine of cleaning and disinfecting is more than sufficient to maintain a safe home environment, even during periods of illness.
Sterilization is reserved for extreme-risk environments, primarily in medical or scientific settings. For example, surgical instruments that enter the body must be sterile. In a residential setting, even after a traumatic event involving biohazards, the highest level of microbial control required is professional disinfection. A certified remediation team like ours will use hospital-grade disinfectants to render the area biologically safe, which is the established standard for restoring a home.
When you are facing a situation that requires more than a standard cleaning solution, the technical requirements can feel overwhelming. 360 Hazardous Cleanup is here to manage the scientific complexities with the compassion and professionalism you deserve, ensuring your property is restored to a verifiably safe state. For immediate, discreet assistance, contact our 24/7 response team.


